Background: The FLT3 inhibitor quizartinib induces quiescence of murine hematopoietic progenitors, and this affords their protection from S phase specific chemotherapies. In this study we investigated whether quizartinib can also protect hematopoietic progenitors from the cytotoxic effects of DNA damaging chemotherapies and radiation.
Methods: C57BL/6 mice were primed with 30 mg/kg of quizartinib or vehicle (5% cyclodextrin) by oral gavage, and treated with DNA damaging chemotherapies, either cyclophosphamide, carboplatin, cisplatin, temozolomide or mitoxantrone 6 hours later, or 5.5Gy total body irradiation (TBI) after 9 hours. This TBI dose represents half the 11 Gy split dose used for bone marrow conditioning of C57BL/6 mice prior to HSC transplantation. Bone marrow cells were analyzed by flow cytometry after 3 to 6 days to identify progenitor and stem cell populations.
Results: In contrast to quizartinib's protection against the cytotoxic effects of 5-FU and gemcitabine, all 5 DNA-damaging chemotherapies caused an increased loss of total bone marrow cellularity when mice were primed with quizartinib compared to vehicle primed controls. Flow cytometry analysis revealed that the numbers of myeloid and multipotent progenitors, and short-term and long-term hematopoietic stem cells (HSCs), were markedly lower in the quizartinib-treated mice.
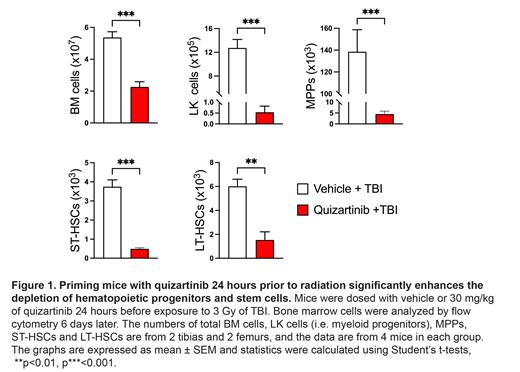
DNA damage and the resultant cell death caused by ionizing radiation is cell cycle specific, particularly for cells traversing from G 1 to S phases of the cell cycle. The ability of quizartinib to induce quiescence in myeloid and multipotent progenitors suggested that quizartinib may protect these cells from radiation. To test this, mice were dosed with vehicle or 30 mg/kg of quizartinib and 9 hours later exposed mice to 5.5 Gy of TBI. Analysis of bone marrow cells at day 6 revealed that total bone marrow cellularity and the numbers of progenitor and stem cells were markedly reduced in quizartinib-dosed mice compared to vehicle controls. For subsequent experiments mice were treated with 3 Gy TBI because the combination of quizartinib and 5.5 Gy caused severe myeloablation. As part of a series of experiments examining the kinetics of quizartinib's sensitization of bone marrow cells we tested whether the sensitization was maintained when mice were dosed with quizartinib 24 hours prior to 3 Gy TBI. Analysis of bone marrow cells 6 days later revealed that quizartinib dosed mice showed a significantly greater loss of total bone marrow cells, progenitor cells and stem cells compared to vehicle dosed mice (Figure 1).
To identify possible mechanisms by which quizartinib sensitizes hematopoietic progenitors to DNA damaging agents we investigated quantitative gene expression profiling of the HPC7 hematopoietic progenitor cell line. These cells provide a suitable in vitro model since quizartinib induces their quiescence and provides protection against 5-FU. HPC7 cells were treated with 100 µM quizartinib, or vehicle, for 18 hr before RNA was harvested and sent for Next-generation sequencing. We found that quiescence induction in HPC7 cells by quizartinib correlated with the transcriptional downregulation of a wide range of genes involved in DNA damage repair pathways (including Brca1/2, Rad51, Palb2, Chek1 and Nbn), thus providing a possible mechanism for the enhanced cytotoxic effects of DNA damaging agents.
Conclusions: Quizartinib sensitizes hematopoietic progenitors and stem cells to DNA-damaging agents, an effect that correlates with the transcriptional downregulation of many DNA damage repair genes. The enhanced depletion of hematopoietic progenitors and stem cells by DNA damaging agents in the presence of quizartinib has important clinical implications. Administering concurrent DNA damaging chemotherapy or radiotherapy with quizartinib treatment should be avoided. However, this interaction could facilitate a reduction in radiation/chemotherapy doses administered for bone marrow conditioning prior to HSC transplantation. By reducing these doses, but not compromising the conditioning process, non-hematopoietic side effects could be minimized and may allow currently excluded patients to be considered for HSC transplantation.
Disclosure: This study was partly funded by Daiichi Sankyo.
Disclosures
Grove:Astellas: Consultancy, Honoraria; Otsuka Australia: Consultancy; Abbvie: Consultancy.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal